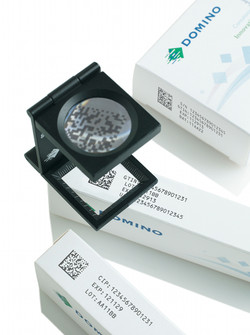
Viewpoint: A shot in the arm for generics producers
Domino inksThere is nowhere to hide from the Falsified Medicines Directive. It impacts generics producers in the same way as any other drug manufacturer or re-packager selling into Europe. With just under 48 months to go before the deadline, which is likely to be mid-2018, the clock is ticking for all producers to get their house in order.
The subject was hot on the agenda at the European Generics Association (EGA) meeting in Madrid earlier this month. As generics manufacturers often supply products with much lower margins than patented drug producers, their budgets are restricted when it comes to new investment – which is why many of them have been holding out until the last minute to find the ‘best-fit’ solution for their businesses.
Related Posts
Revenue officials seize 45,000 cigarettes at Dublin Airport
Revenue officers have seized 45,000 cigarettes at Dublin Airport. The seizure...
Fake washing detergent making factory busted in Delhi, 1 held
New Delhi, Mar 21 (PTI) The Delhi Police has busted a factory that was...
Cops seize over Dh100 million worth of fake goods in UAE.
04, November 2018 | Khaleej Times More than Dh100 million worth of...
Authorities fear illicit tobacco trade is moving from Gloucester shops to ‘tab houses’ on estates
Fears have been raised that the war on illicit tobacco could soon take a sinister...





